Use Case 1 Validation & Evaluation Workshop on Lung Cancer (Phase 2), 25 February 2026
Nottingham Trent University organised the Use Case 1 Validation & Evaluation Workshop for Phase 2 dedicated to lung cancer research, bringing together project partners and external stakeholders to review progress and demonstrate newly developed tools and datasets supporting the project’s Health Data Hub.
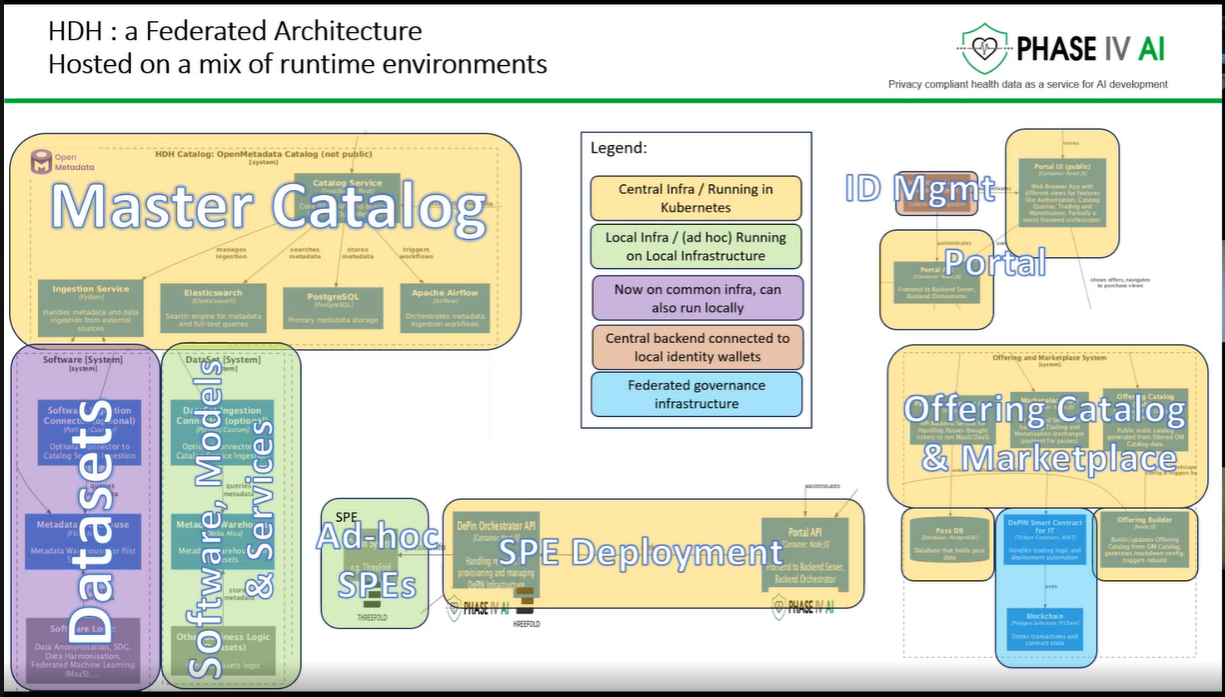
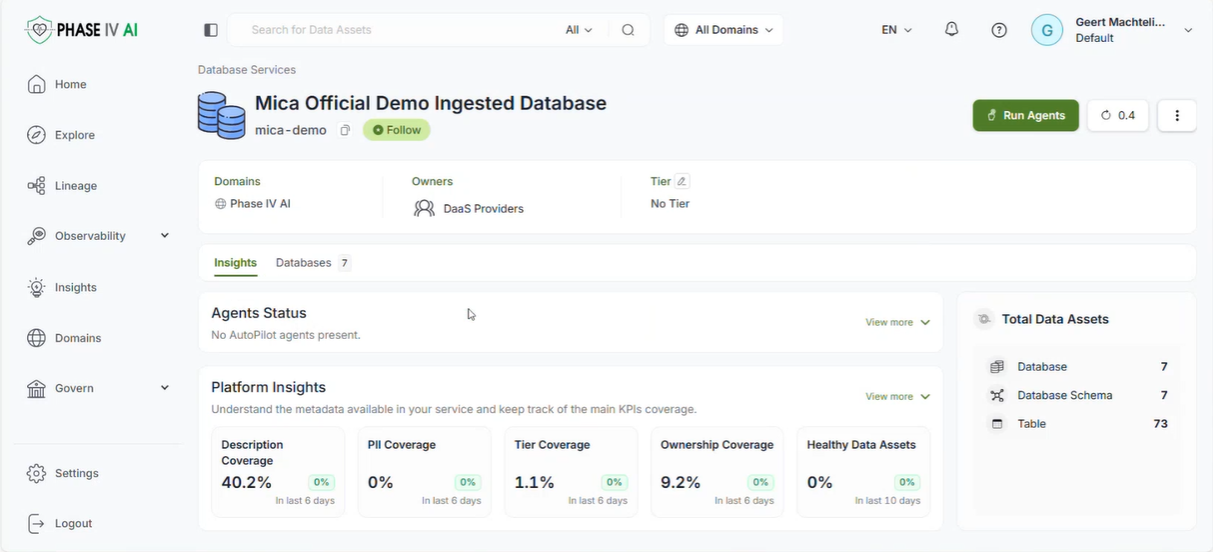
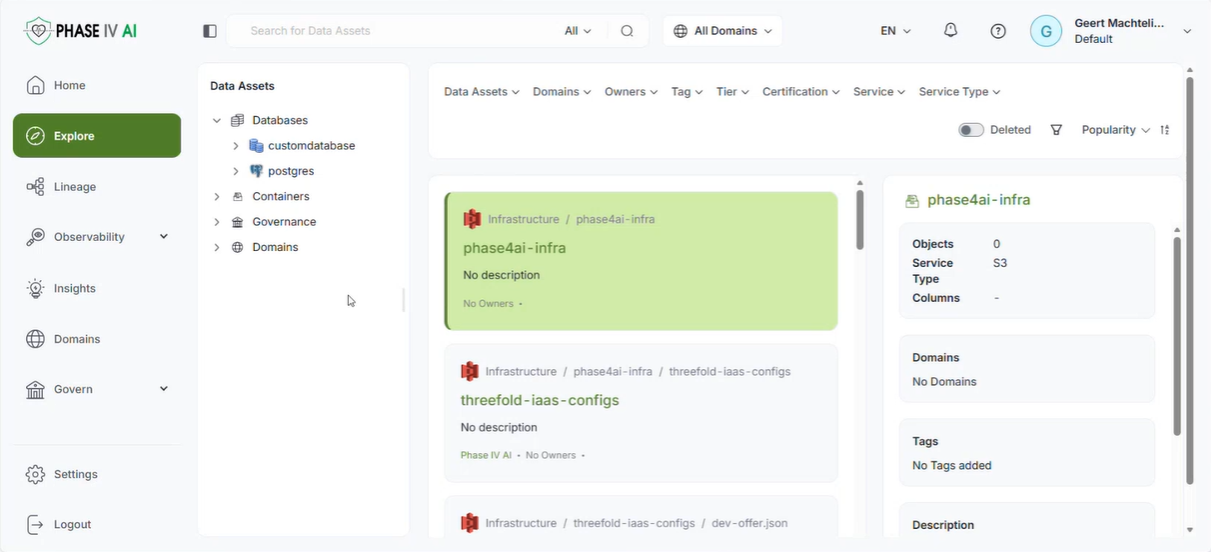
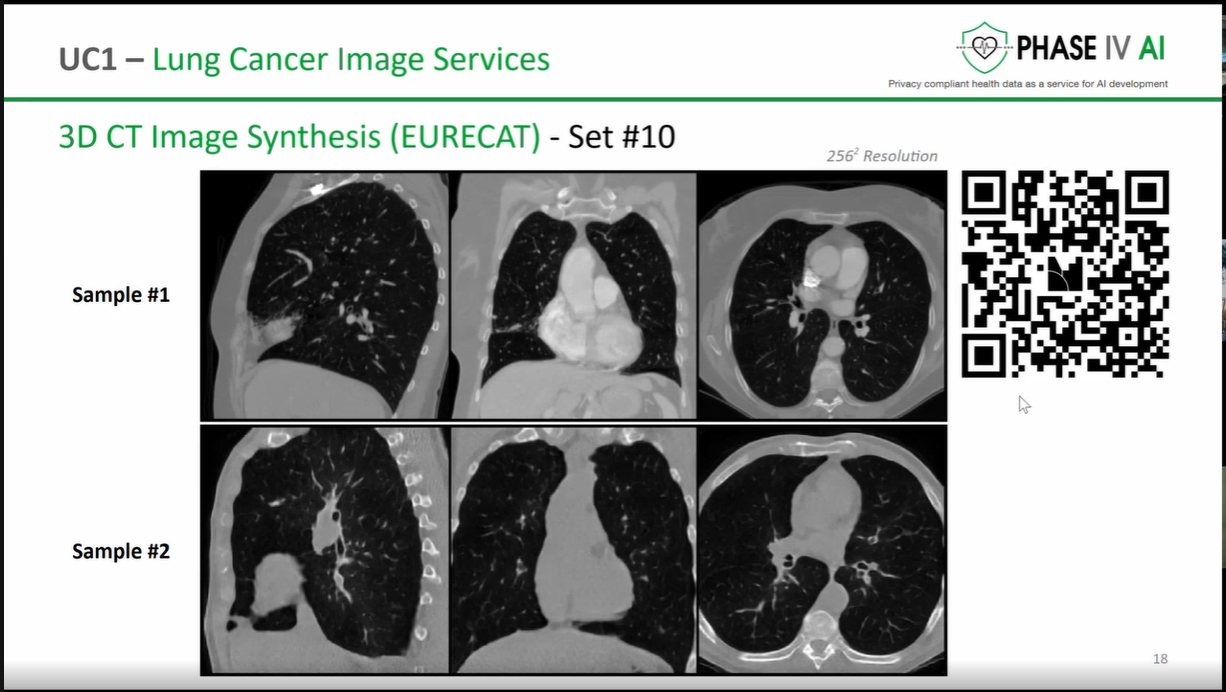
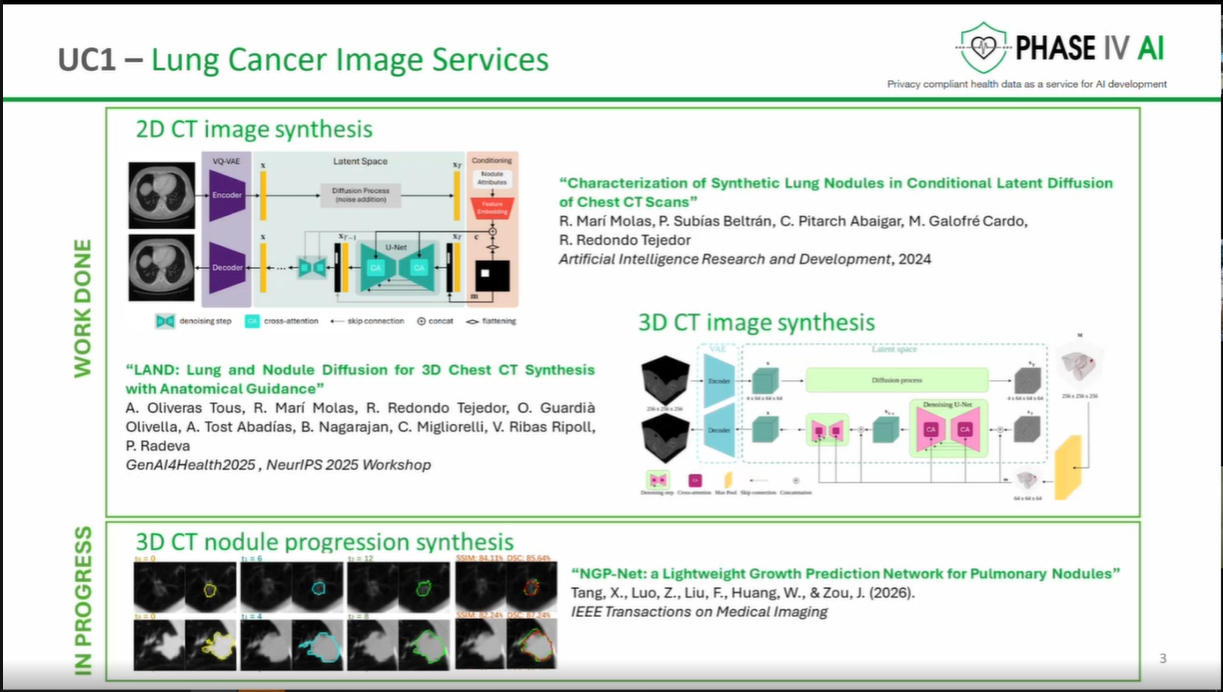
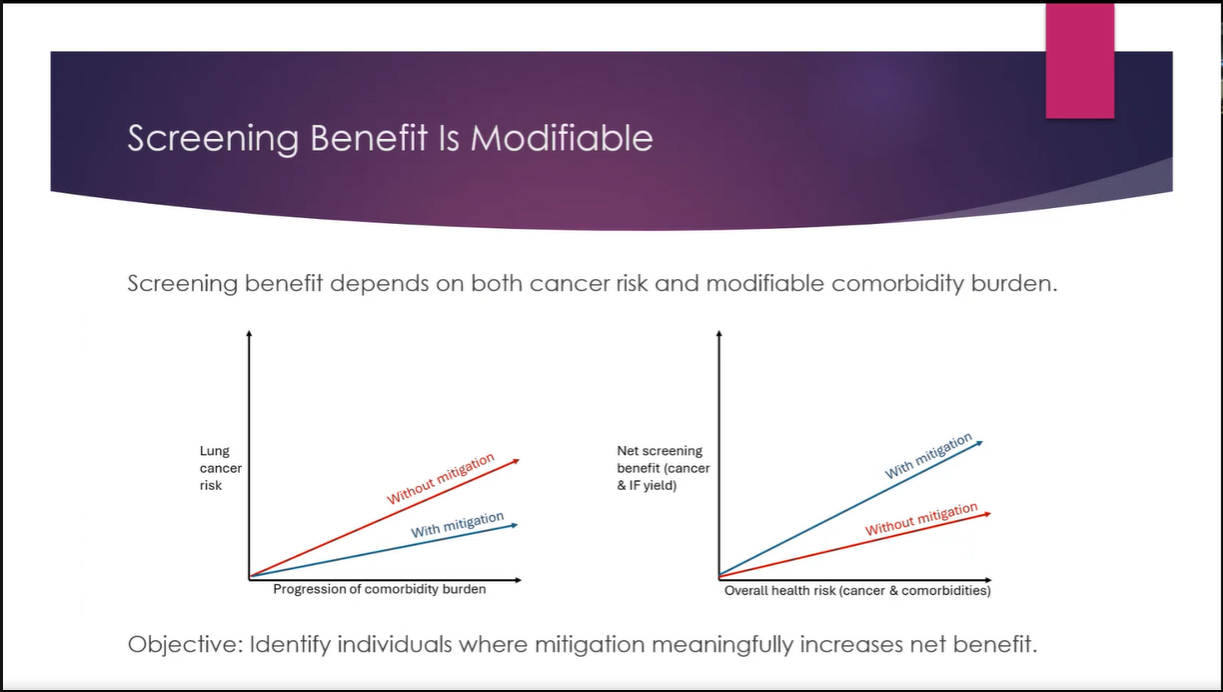
The workshop provided an opportunity to showcase the latest advancements in the cataloguing, harmonisation, and generation of health data, as well as prototype technologies designed to support lung cancer research and risk prediction. Several partners presented technical developments that contribute to the project’s goal of enabling secure, interoperable, and privacy-preserving health data sharing across Europe.
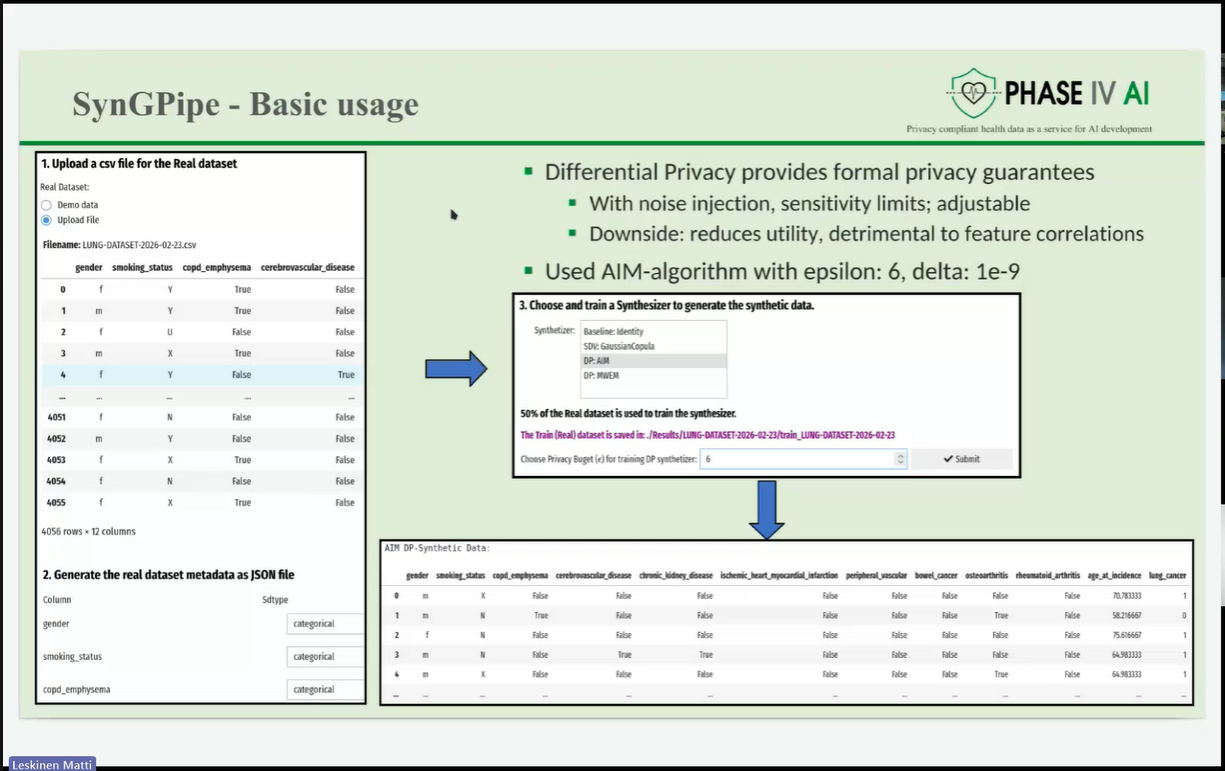
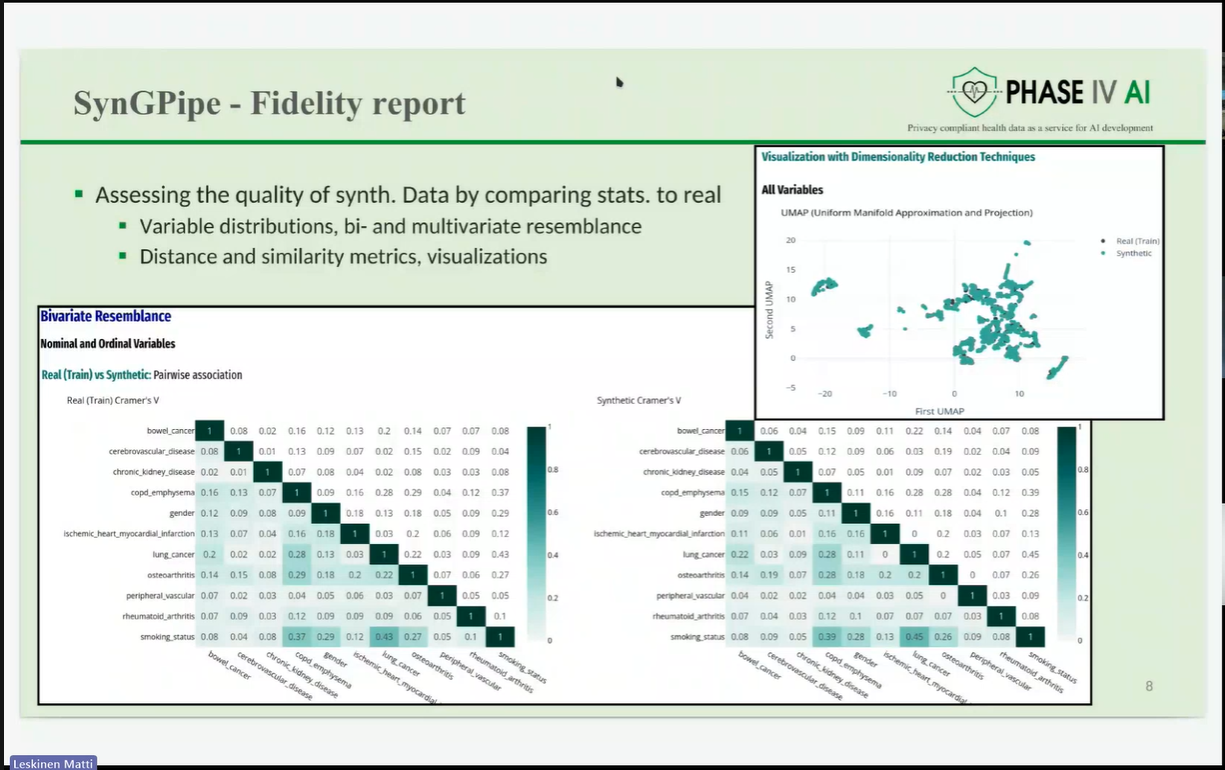
The session opened with an introduction to the PHASE IV AI project, followed by a presentation on the Health Data Hub catalogues, models, datasets, and harmonisation tools. Partners also introduced a synthetic lung imaging dataset, demonstrating how synthetic data can support research and model development while protecting patient privacy. During the second part of the workshop, partners presented planned work on CPRD lung electronic health record (EHR) data, as well as the Syn-G-Pipe prototype, a pipeline-based tool designed to facilitate synthetic data generation workflows. Additional presentations focused on lung cancer EHR models and a prototype Lung Cancer Risk Prediction Tool, which was demonstrated in an interactive session.
The workshop concluded with a summary discussion and feedback session, allowing partners to exchange insights and provide input for the next stage of development (Phase 3). These collaborative validation activities play an important role in ensuring that the tools and datasets developed within the project meet the needs of healthcare research.